💡 Ebola can now be cured - this is how it works
Two new treatments can treat Ebola effectively. From now on, we will no longer say that Ebola is incurable, says the person responsible for the study.
Share this story!
In the midst of a chaos of violence and war, researchers and doctors in the Democratic Republic of Congo have conducted a clinical study of new drugs to try to combat the Ebola outbreak that has lasted for a year.
On Monday, the study's sponsors, the World Health Organization WHO and the National Institutes of Health , announced that two of the experimental treatments appear to increase survival rates dramatically.
We have previously reported on an experimental Ebola vaccine that has been shown to protect people from getting the disease. The treatments that have now been developed show that it is possible to cure people who have already been infected.
Jean-Jacques Muyembe, director general of the Congo's Institut National de Recherche Biomédicale in the Democratic Republic of Congo, who was in charge of the study, told Reuters that the results meant that "from now on we will no longer say that Ebola is incurable."
The study, which aims to develop these treatments, started in November 2019. At that time, the treatments were given to a random sample of patients in four treatment centers in the eastern parts of the country, where the outbreak is at its worst.
The treatments were one of four different variants: an antiviral drug called remdesivir or one of three drugs that use monoclonal antibodies. One of these, a drug called ZMapp, is currently the standard treatment for Ebola outbreaks. ZMapp had been tested and used during the devastating Ebola epidemic in West Africa in 2014 and the goal was to see if these other drugs could surpass it. Preliminary data from the first 681 patients, 725 planned, showed such strong results that the study has now been completed.
Patients who received Zmapp in the four trial centers had a total mortality rate of 49 percent, according to Anthony Fauci, director of the NIH's National Institute of Allergy and Infectious Diseases. For infected individuals who do not seek any form of treatment, the mortality rate exceeds 75 percent.
The monoclonal cocktail of antibodies produced by Regeneron Pharmaceuticals had the greatest effect on reducing mortality, down to 29 percent, while NIAID's monoclonal antibody, called mAb114, reduced mortality to 34 percent.
The results were most striking for patients receiving treatment shortly after they became ill, when their viral load was still low. Here, mortality decreased to 11 percent with mAb114 and to 6 percent with Regeneron's drugs, compared with 24 percent with ZMapp and 33 percent with Remdesivir.
A new study will now be launched where Regeneron's treatment will be compared with mAb114, which is produced by a Florida-based company called Ridgeback Biotherapeutics. All treatment units in the outbreak area will now only administer the two most effective monoclonal antibody drugs, according to WHO's Mike Ryan.
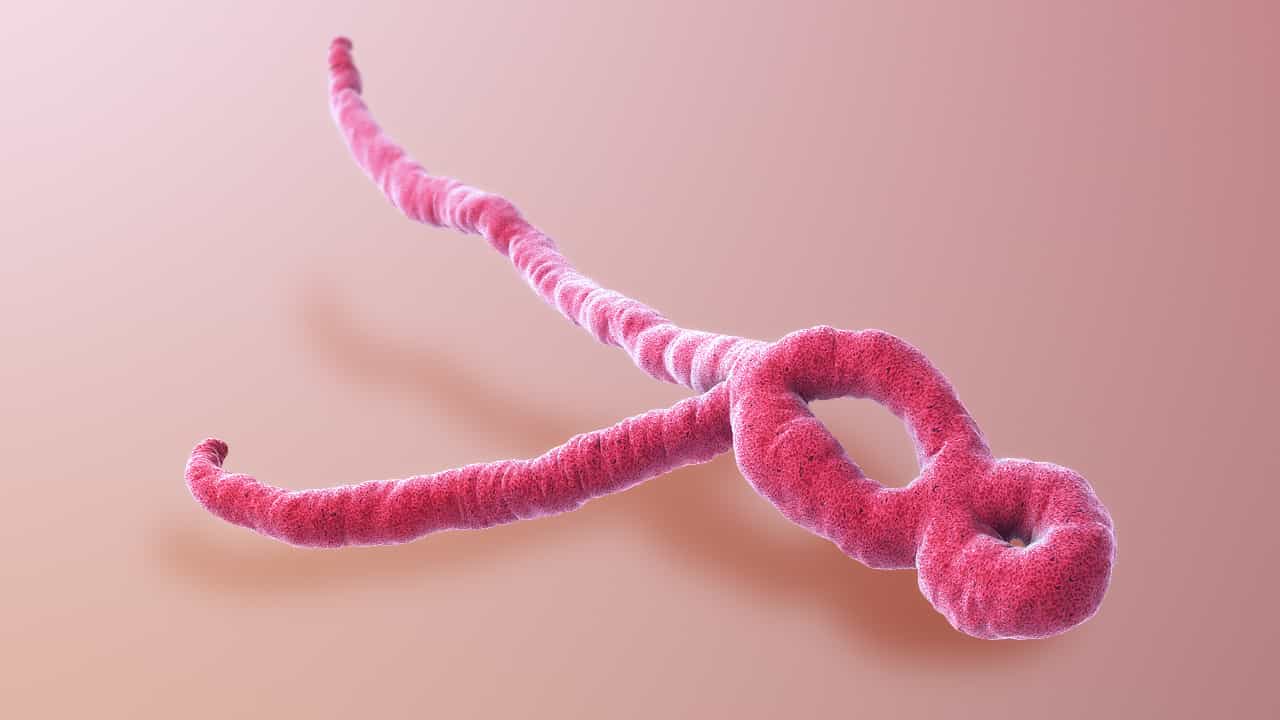
Image: A 3D medical animation still of Ebola virus, Wikimedia.
By becoming a premium supporter, you help in the creation and sharing of fact-based optimistic news all over the world.