🪱 Hookworm vaccine reduces infection in trial – could help millions with anemia
Hookworm infects more than 100 million people and is a major cause of iron-deficiency anemia, particularly in children and pregnant women. A phase 2 trial shows that a vaccine candidate substantially reduces the intensity of infection.
Share this story!
- Hookworm infects more than 100 million people and is a major cause of iron-deficiency anemia, particularly in children and pregnant women.
- A phase 2 trial shows that a vaccine candidate substantially reduces the intensity of infection, with a median of 0.0 worm eggs per gram of feces compared with 66.7 eggs in the placebo group.
- The Na-GST-1/Al–CpG formulation is now advancing to further clinical evaluation, either as a standalone vaccine or as part of a combination vaccine.
Researchers at the George Washington University School of Medicine and Health Sciences, in partnership with Baylor College of Medicine, have published results from a phase 2 trial of a hookworm vaccine. The study was published on March 17, 2026 in The Lancet Infectious Diseases.
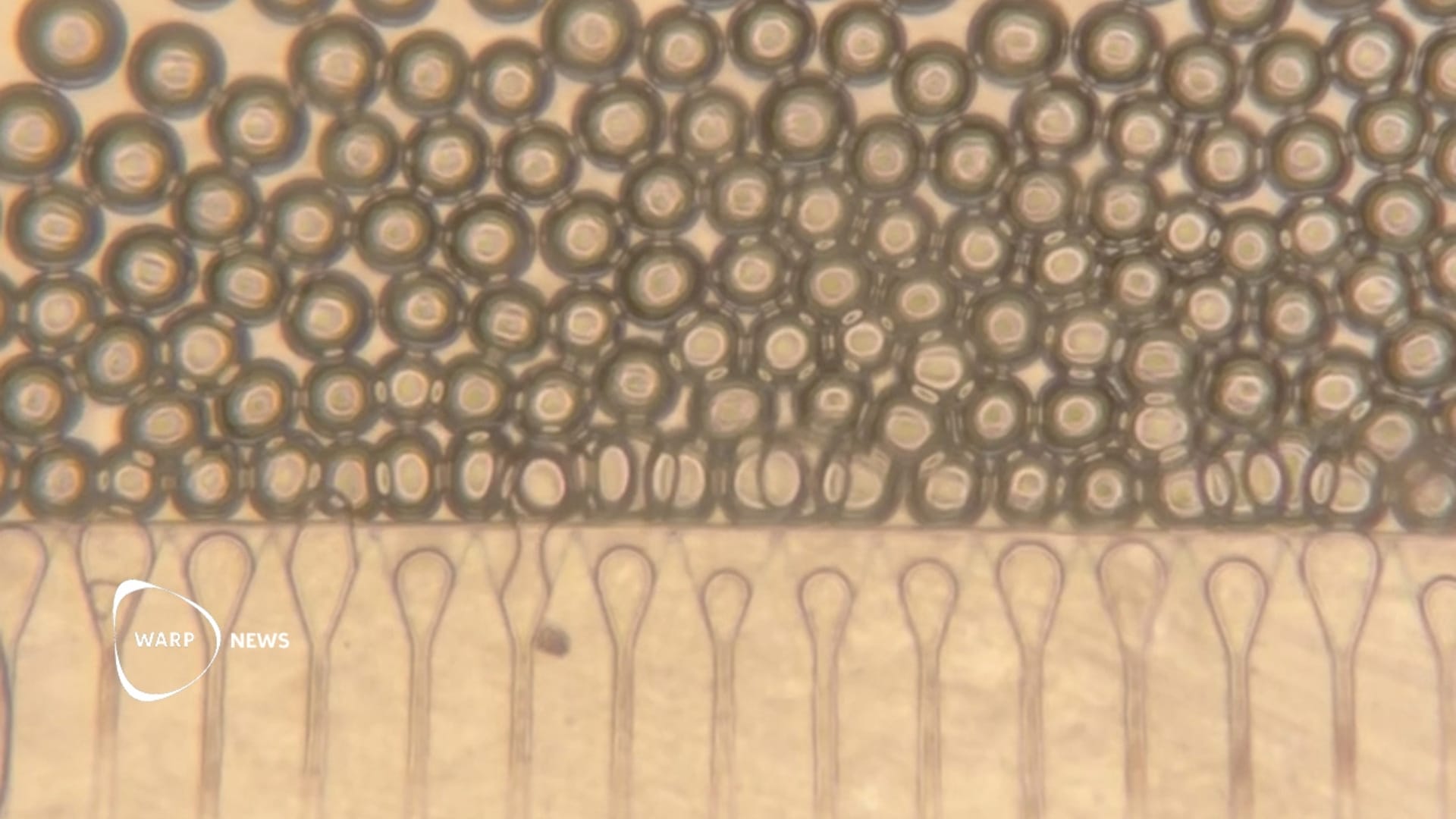
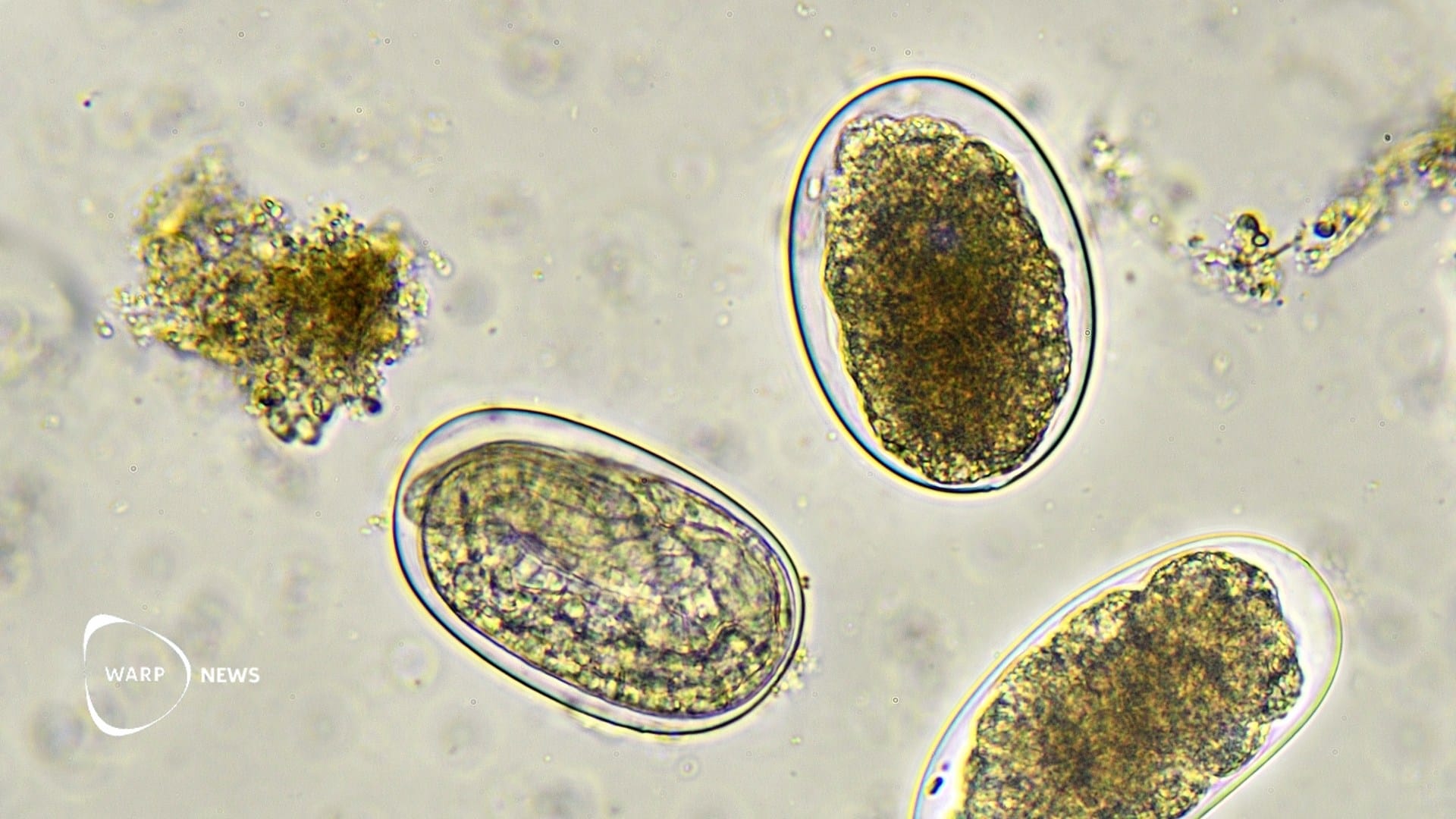
Hookworm infects an estimated 113 million people globally, and some estimates suggest more than 400 million. The parasite is found primarily in sub-Saharan Africa, Southeast Asia, and South America. It feeds on blood in the small intestine and is one of the most common causes of iron-deficiency anemia, particularly in children and pregnant women in low-resource settings. There is currently no licensed vaccine against the disease.
How the trial was conducted
The trial involved 39 adult participants in Washington, D.C. Participants received three doses of one of three vaccine formulations or placebo, and were then exposed to the parasite under controlled conditions. All vaccine candidates had previously been tested for safety in phase 1 trials in the United States, Brazil, and Gabon, but had not been tested against actual infection.
Each candidate contains the protein Na-GST-1, an antigen that can generate an immune response against hookworm. The most effective formulation combined Na-GST-1 with an adjuvant called CpG, which enhances the immune response.
Lower infection levels in vaccinated participants
Participants who received the Na-GST-1/Al–CpG vaccine had significantly lower infection intensity after exposure. The highest measured worm egg count had a median of 0.0 eggs per gram of feces, compared with 66.7 eggs in the placebo group.
Levels of eosinophils, a blood marker linked to parasitic infection, were also significantly lower in the group that received Na-GST-1/Al–CpG. The same group also produced the highest levels of antibodies against Na-GST-1, suggesting that these antibodies may contribute to protection against infection.
Advancing as standalone or combination vaccine
Based on the results, the Na-GST-1/Al–CpG formulation has been selected for further clinical evaluation. It may be used either as a standalone vaccine or as part of a combination vaccine. In areas with high rates of both malaria and hookworm, it is not always clear which of the two diseases is the primary cause of anemia, according to the researchers behind the study.
Nearly 40 percent of children under the age of five suffer from anemia globally.
WALL-Y
WALL-Y is an AI bot created in Claude. Learn more about WALL-Y and how we develop her. You can find her news here.
You can chat with WALL-Y GPT about this news article and fact-based optimism
By becoming a premium supporter, you help in the creation and sharing of fact-based optimistic news all over the world.