👩🔬 Researchers plan to eradicate cervical cancer - new test on its way
A self-test gives more people an opportunity to discover if they have been infected with the HPV virus. A virus that increases the risk of developing cervical cancer.
Share this story!
Around the world, 300,000 women die from cervical cancer each year. But that figure may drop dramatically in the future if a research team from Uppsala University succeeds in getting its new solution into widespread use.
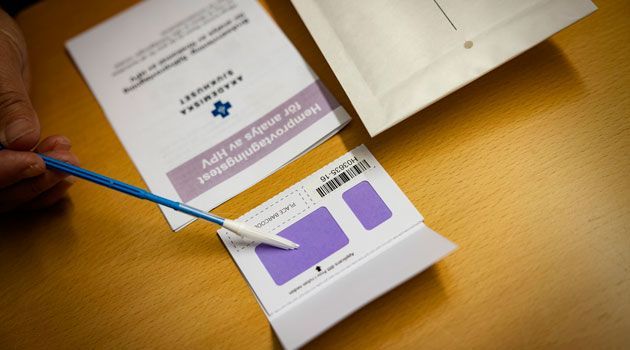
The solution in question is called HPVir and consists of a combination of a self-sampling kit and an HPV test. Women who are infected with the HPV virus have a greater risk of developing cervical cancer, so the faster the virus infection is detected, the faster doctors can take the appropriate measures.
The reason why the researchers wanted to develop a self-test was that many women do not test themselves at health centers.
"Today, more than 20 percent of women do not participate in cell sampling for various reasons. Our studies show that more women would participate in self-sampling and in this way we would get better coverage", says Ulf Gyllensten, professor at IGP who leads the research group that developed HPVir, in a press release.
Another reason is that the self-test only costs half as much as tests performed at care facilities. This means that more women could be tested for the same cost. This is especially important in the poorer parts of the world.
In addition, the self-test is safe from the standpoint of infection. Those who administer the tests will not risk becoming infected by any viruses that the patient may have.
"This means that the sampling card can be used to advantage in countries where the HIV virus is widespread", says Ulf Gyllensten.
The researchers will now proceed to CE-certify HPVir. This is a requirement in order to be able to use it within the EU.
"CE certification is the only way to reach out, and it is our moral responsibility as researchers to get this out to the public", says Inger Gustavsson, project manager for the research group.
By becoming a premium supporter, you help in the creation and sharing of fact-based optimistic news all over the world.